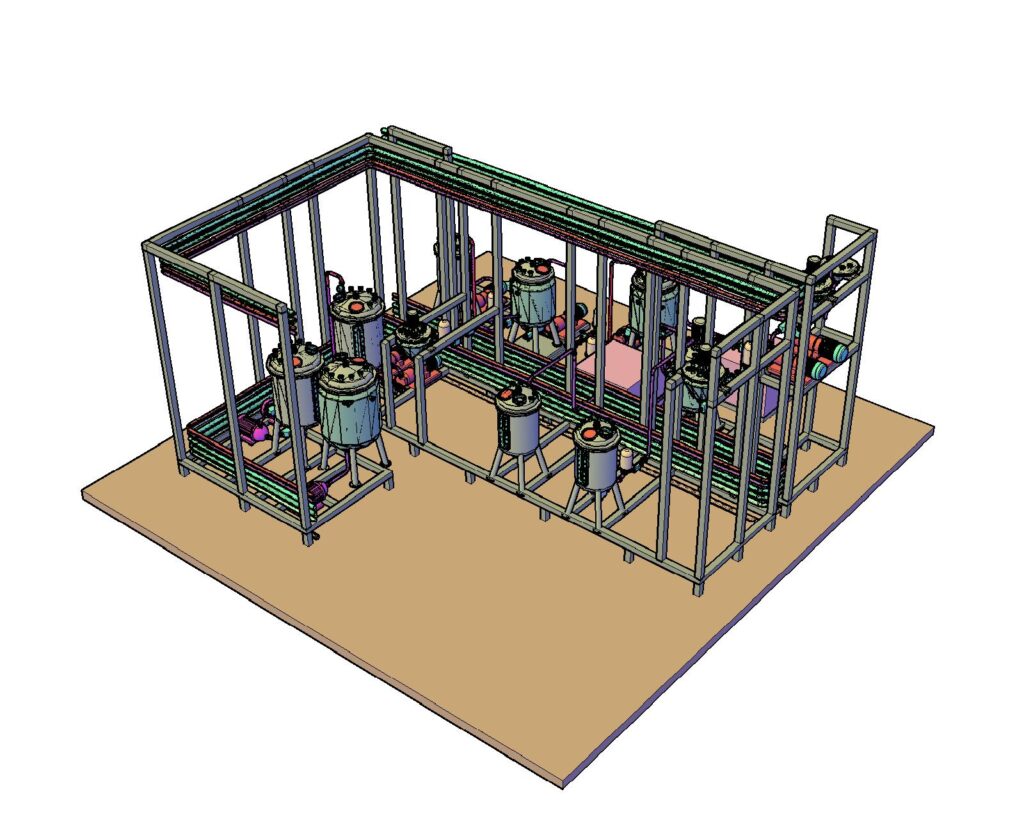
Processing Plant for Liquid Parenteral

Parenteral Processing Plant
We offer Turnkey Solutions for Sterile Liquid Processing.
Sterile Skids for Manufacturing, Filtration, Storage and Holding areas and also Buffer Vessels with Integrating
Piping for Sterile product transfer, Piping for Upstream /downstream Integrating them with Centralised CIP/ SIP
with full Automation to 21CFR and beyond.
We offer customised Sterile Processing plant solutions for Pharmaceuticals And Biotech Achieving desired standards, Step by Step:
- Discussing P&ID with users requirement
- Designing and manufacturing vessels skids for Parenteral Manufacturing, Filtration, Storage and Holding areas and for Upstream / Downstream
- Piping and centralized CIP/ SIP with automation required by client up to 21 CFR and beyond.
- IUS Equipment could be among the best as a single source to manufacture supply execute with the highest automation Level.
Scope of Application
PHARMACEUTICALS
- Vials
- Ampoule
- V. fluids
- Aerosols
- LVP
BIO PHARMACEUTICALS
- Vaccines
- Human Insulins
- Serums
- Plasma Fractionations
- Cell culture
We also manufacure and supply Independent Sterile Vessels with Capacity: 1 Ltr TO 5000 Ltr. And Designed as per CGMp.
Manufacturing – Filtration Skids
DESIGNING HIGHLIGHTS
- Automation in compliance with 21CFR Part 11.
- Vessels are CIP/SIP able.
- Bottom as well as top driven stirrer & high shear mixer.
- 100% Drainability.
- Contact part electropolished upto 0.3 Ra.
- Load cell for precise transfer
- Complete FAT & documentation.
- Clean-In-Place (CIP) & Sterilization-In-Place (SIP) Systems
- Sanitary design to meet all cGMP criteria.
- Contact parts: SS 316 L, mirror polished & EP.
- All non-contact parts: SS 304 Matt finish.
- Orbital tube welding.
- Sanitary moisture separator & pressure regulating valve.
- Automation as per USFDA 21 CFR Part 11.
- Built to custom requirement.
- Cleaning validation with riboflavin test.
- 100% Drianability.
- GMP documentation like DQ, FAT, IQ, OQ & PQ protocols.
- Site installation & commissioning
Documented output of cleaning & sterilization cycles.
Interconnecting Piping
- Inter-connected Pipings / Valves / Instrumentation.
- SS 304 polished skid.
- Automation System/Control Panel.
- PLC Control Panel with Time / Temperature control program, printer, alarm, etc. as required
Clean-in-place (Cip) & Sterilization-in-place (Sip) Systems:
Cleaning-In-Place (CIP) and Sterilization-In-Place (SIP) are systems designed for automatic cleaning and disinfecting. We design, develop, manufacture, supply, and install Mobile and Fixed CIP & SIP Units for
sanitization and sterilization. The units are custom made, modular, skidded in automated or semi-automated Models as per the required time cycle for cleaning and Sterilization of portable to large fixed Multi-Tank system Sterilization of portable to large fixed Multi-Tank system Tank/ Tank less system.
Automation Solution
Designed to monitor, control, achieve and analyse the process data. Our control systems are of the highest standards of compliance to various regulatory guidelines. Thus our integrated approach for your process needs relieves you of approaching multiple agencies to get your plant started and running.
All this has been achieved through our continuous perseverance, hard work, research in product design, adherence to quality and our total commitment to business ethics. The determined efforts of our dedicated team have gained us global reputation & reliability.
3D & Photo Gallery